
 |
| Home>iPhone>Drug Alert |
| Drug Alert - Side Effects and Overdose Symptoms in Plain English
View and File FDA Adverse Event Reports / View the Latest FDA Drug Safety Info |
Click here to contact us about this App
| The first time you start Drug Alert there will be a short delay while the drug information is prepared. This is normal. | |
| Disclaimer | |
| The Drug List: | Use the index and scroll to find a particular drug. Recalled and discontinued drugs are labeled: |
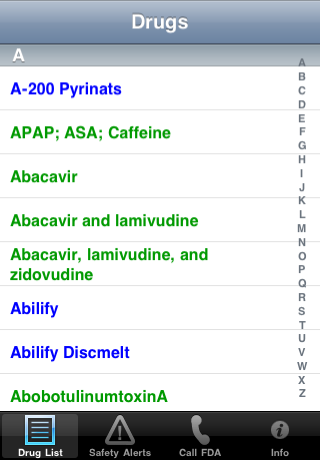 | 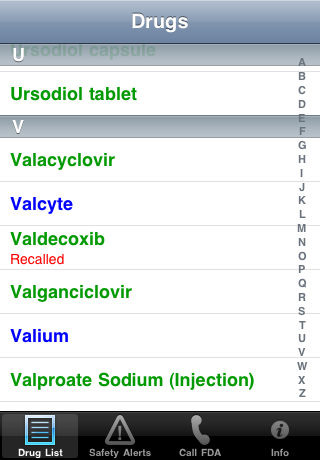 |
| Touch a Drug to See Options and Generic Name: | Side Effects: |
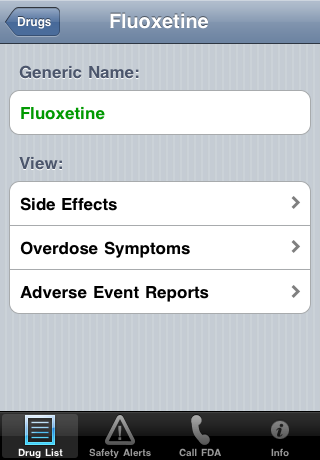 |  |
| Overdose Symptoms: | |
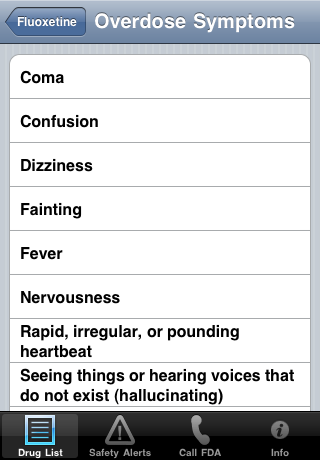 | |
| Adverse Event Reports | |
| An adverse drug reaction is any undesirable experience associated with the use of a medicine in a patient. Adverse Event Reports
are reports of adverse drug reactions. These reports can and should be be filed by anyone who has experienced an adverse drug reaction or knows someone who has. | |
| Adverse Event Reports have specific outcomes: Death, Life Threatening Condition, Hospitalization, Disability, Congenital Abnormality
(Birth Defect), Required Intervention to Prevent Permanent Impairment/Damage and Other. An Adverse Event may have more than one outcome. For example, the person may have been hospitalized and then died. In addition, a drug may be assigned to an adverse event report as a primary suspect, a secondary suspect, concomitant (occurring at the same time) or interaction. | |
| Drug Alert presents a summary of Adverse Event Reports for any selected drug. The summary is based on the most recent year of available
reports. The summary includes only those reports where the drug was listed as the primary suspect in the adverse event. Only the 30 most reported reactions are shown. | |
| NOTE: An Internet connection is required to see the Adverse Reaction Report summaries. | |
| Touch Adverse Event Report on the Options screen. The number of reports and time period are shown: | Below that you see the outcomes and how many reports had that outcome: |
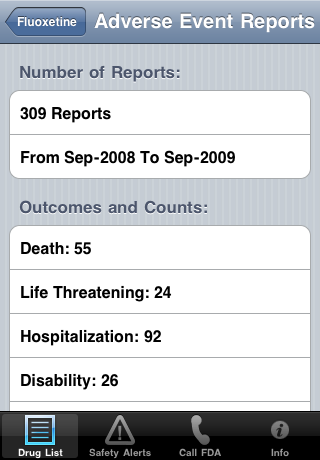 |  |
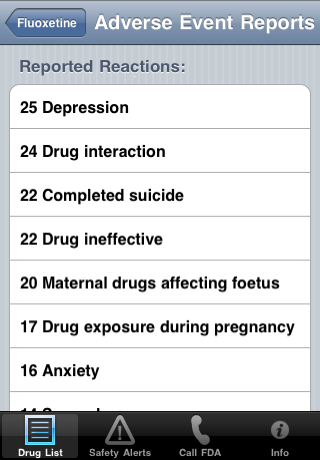
| Below the outcomes are the 30 most reported reactions and how many where reported: | |
 | |
| File An Adverse Event Report | |
| You can file an Adverse Event Report directly from the App by calling the FDA. The report can be for yourself or someone
you know. The patient's name does not have to be given. You do need to have a list of all the drugs the patient is taking, how long they have been taking them, the dosages, how often taken and by what method (orally, injection, etc.) Touch the "Call FDA" button at the bottom of the screen and the then "Call The FDA" button: | |
 | |
| FDA Drug Safety Alerts | |
| Drug Safety Alerts are issued by the FDA to inform consumers of drug safety issues. The Safety Alerts are
available on the FDA web site. You can view the FDA Safety Alert web site directly from the App. | |
| NOTE: An Internet connection is required for this feature. | |
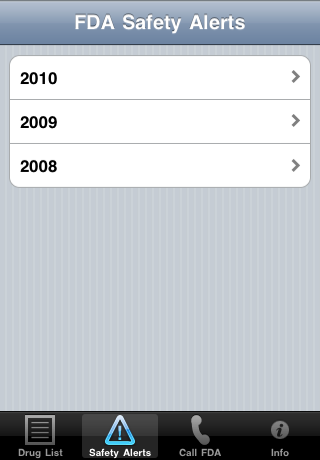
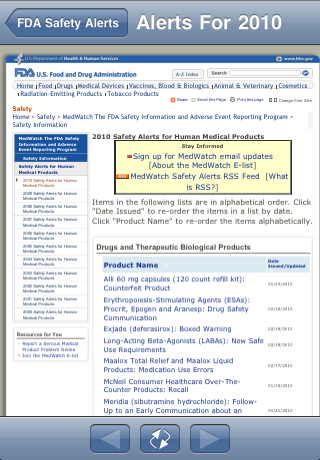
| Touch the "Safety Alerts" button at the bottom of the screen: | Touch a year. The FDA Web site is shown. You can navigate in the web site. Use the left and right arrow buttons for "Back" and "Forward" |
 |  |
| You can also view and navigate in landscape orientation: | |
 | |